 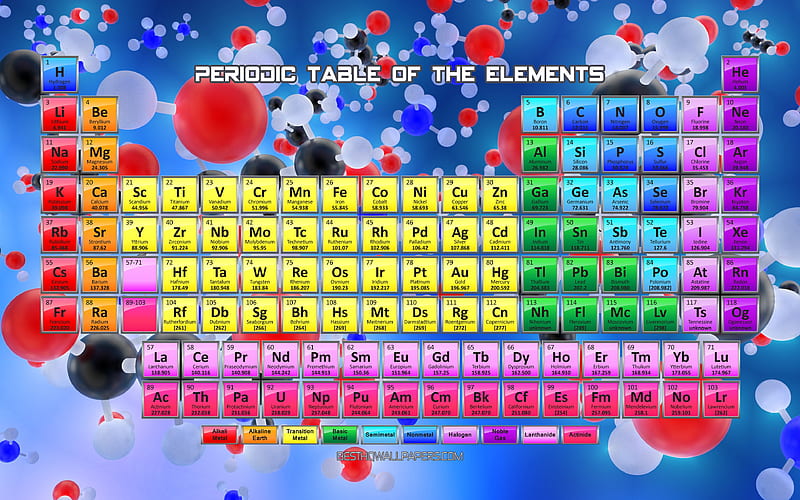
It is a physical science within the natural sciences that studies the chemical elements that make up matter and compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during reactions with other substances. Periodic trends, arising from the arrangement of the periodic table, provide chemists with an invaluable tool to quickly predict an element's properties. Chemistry is the scientific study of the properties and behavior of matter. Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element, including its size and its electronic properties. 2.5: Some Characteristics of Different Groups The periodic table is useful for understanding atomic properties that show periodic trends.Some characteristics of the elements are related to their position on the periodic table. 2.4: The Periodic Table The chemical elements are arranged in a chart called the periodic table.Atoms have a mass that is based largely on the number of protons and neutrons in their nucleus. This is determined based on the number of electrons that would be added, lost, or shared if it reacts with other atoms. If you learned in chemistry that some atoms tend to gain or lose electrons or form bonds with each other, those facts remain true even when the atoms or molecules are part of a living thing. Valence describes how easily an atom or radical can combine with other chemical species. Atoms and molecules follow the rules of chemistry and physics, even when theyre part of a complex, living, breathing being. Isotopes are atoms of the same element that have different masses. The words valence and valency have two related meanings in chemistry. 2.3: Isotopes and Atomic Weight Elements can be identified by their atomic number and mass number.Atoms or ions with the same electronic configurations are said to be. Isoelectronic chemical species typically display similar chemical properties. The term means 'equal electric' or 'equal charge'. Protons and neutrons are grouped together in the nucleus of an atom, while electrons orbit about the nucleus. Isoelectronic refers to two atoms, ions, or molecules that have the same electronic structure and the same number of valence electrons. 2.2: Elements and Atomic Number Atoms are composed of three main subatomic particles: protons, neutrons, and electrons.The modern atomic theory establishes the concepts of atoms and how they compose matter. Diphosphorus (P 2 ), disulfur (S 2 ), dilithium (Li 2 ), and disodium (Na 2 ) all occur when their solids are evaporated, but these diatomic elements only exist as gases that repolymerize when cooled. 2.1: Atomic Theory and the Structure of Atoms Atoms are the ultimate building blocks of all matter. However, these molecules are not very stable, so their chemical bonds are readily broken. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
